 |
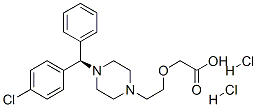 |
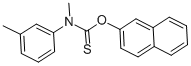 |
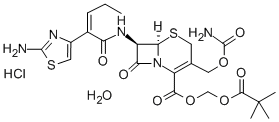 |
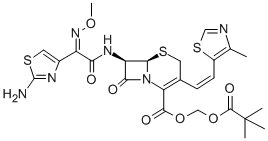 |
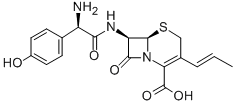 |
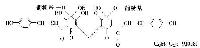 |
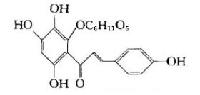 |
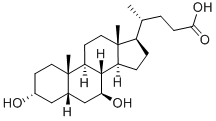 |
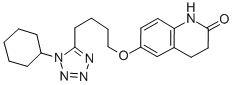 |
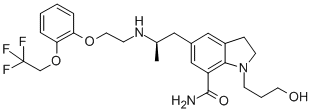 |
 Info marked by "
Info marked by "
Overview
Our history
Zhejiang Yongning Pharmaceutical Co., Ltd., whose predecessor is Zhejiang Yongning Pharmaceutical Factory (established in 1974), has a total of 1200 staff, including 180 tec...
View More >
 Zhejiang Yongning Pharmaceutical Co., Ltd.
Zhejiang Yongning Pharmaceutical Co., Ltd.